Supply Chain for Prescription Medications Mitigating Risks from Human Xylazine Exposure Public Meeting
Advancing Psychedelic Clinical Study Design Prescription Stimulant Availability
Considerations for Buprenorphine Initiation and Maintenance Care Understanding Fatal Overdose
Stimulant Use Disorder Treatment Workshop Harm Reduction Roundtable
Individual & Provider Perspectives Naloxone Access Payor Perspectives Ketamine Therapeutic Use
The United States is facing an overdose crisis. Over 107,000 Americans died of drug overdoses in 2021, according to the Centers for Disease Control and Prevention’s provisional estimates. According to the Department of Health and Human Services (HHS), the overdose crisis has evolved over time and is now  largely characterized by deaths involving illicitly manufactured synthetic opioids, including fentanyl, and, increasingly, stimulants. Since 1999, the rate of overdose deaths has increased by more than 250%. In response to these changes, HHS launched its Overdose Prevention Strategy to focus on primary prevention, harm reduction, evidence-based treatment, and recovery support.
largely characterized by deaths involving illicitly manufactured synthetic opioids, including fentanyl, and, increasingly, stimulants. Since 1999, the rate of overdose deaths has increased by more than 250%. In response to these changes, HHS launched its Overdose Prevention Strategy to focus on primary prevention, harm reduction, evidence-based treatment, and recovery support.
The Reagan-Udall Foundation for the FDA (FDA Foundation) supports the U.S. Food and Drug Administration’s work to develop strategies for the prevention, treatment, and management of substance use disorders by engaging with stakeholders in the health, regulatory, industry, caregiver and advocacy communities to identify and address critical gaps across the SUD treatment system.
Demand Modeling Assessment
Each year, the Food and Drug Administration (FDA) submits an estimate of medical, scientific, and reserve stock needs for Schedule I and II substances to the Drug Enforcement Administration (DEA). In turn, DEA uses these estimates, along with data from other sources, to set an overall quota or limit for these substances, as well as individual quotas for manufacturers of these substances. To inform potential improvements in the quality of these annual estimates, the Foundation is conducting an analysis on behalf of the FDA to evaluate forecasting techniques used by government agencies, the private sector, and academic researchers to predict demand for controlled substances. If you are interested in learning more about the project or contributing to this analysis, please fill out this form to receive updates.

The Foundation is collaborating with FDA to explore the current landscape and potential opportunities for safe disposal methods for unused and expired opioid medications. The project will focus on understanding consumer perspectives and needs related to opioid disposal.
The FDA's fifth Online Controlled Substances Summit was convened by the Foundation on July 26, 2024. The meeting focused on identifying innovative solutions to reduce the availability of controlled substances online. The public meeting featured FDA leadership and other experts as they examined the impact and evolution of online purchases of controlled substances. In addition to the public meeting, the Foundation convened a closed working session including social media, internet, technology, and controlled substances experts working collaboratively to identify immediately implementable actions and long-term strategies aimed at disrupting the trajectory of the current crisis.
The Foundation hosted a public meeting titled "Advancing Treatments for Post- Traumatic Stress Disorder," in alignment with the FDA's ongoing efforts to better understand the medical needs surrounding PTSD with a goal of facilitating treatment development. This meeting included a panel discussion with federal partners exploring efforts to accelerate treatment development for PTSD, including the potential role of psychedelic drugs. Following the panel, stakeholders were invited to share their perspectives, both in-person and virtually. A diverse range of voices contributed, including individuals with lived PTSD experience, their families, patients, advocates, researchers, scientists, and drug developers.
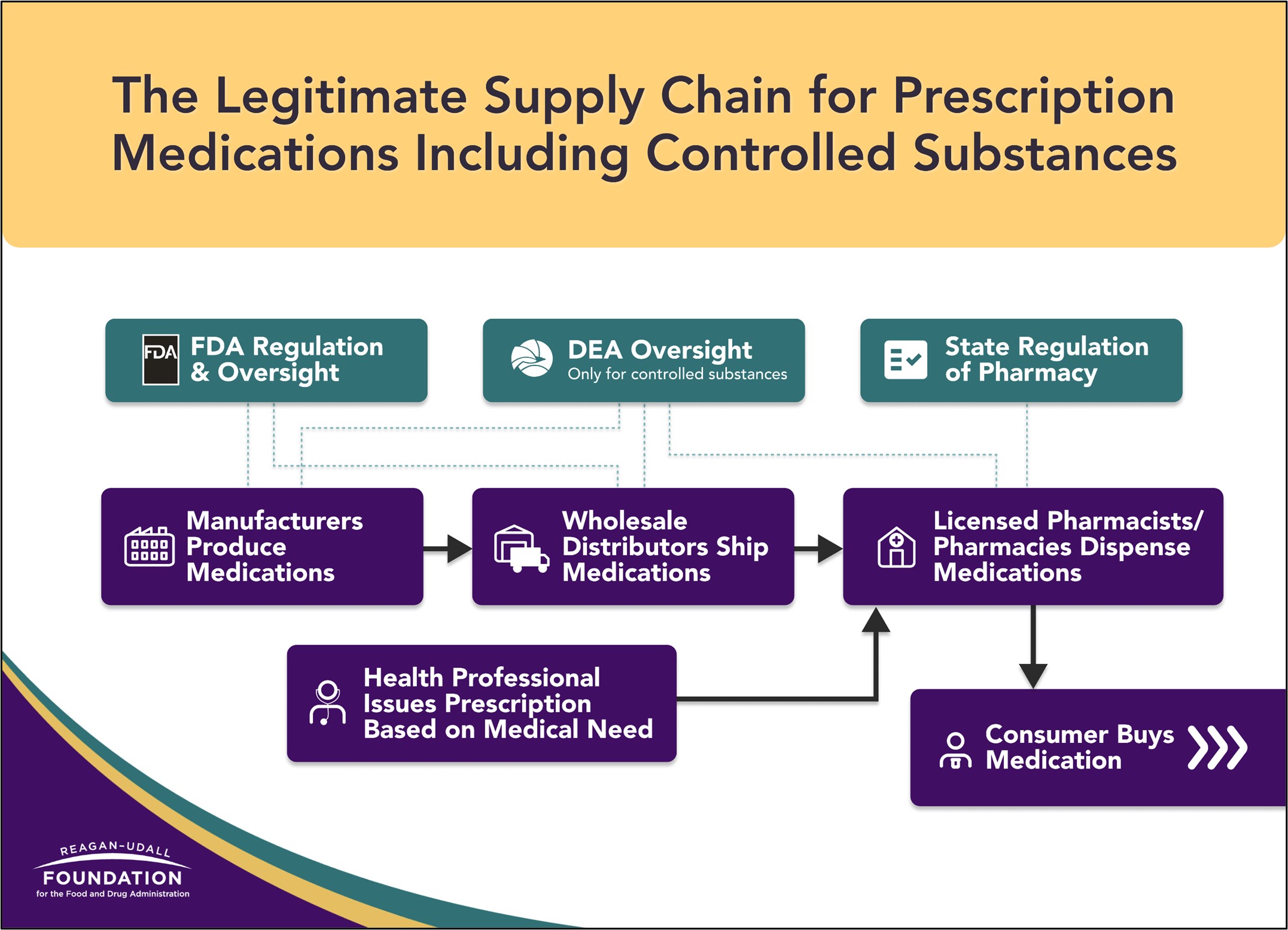
As people turn toward telehealth, accessing care through video calls or app-based services, they are also turning online to access medications. Purchasing controlled substances and other prescription medications online may occur through licensed pharmacies, but also via illegal online pharmacies or non-licensed sellers on websites, social media or apps—often without the consumer even realizing. Differentiating between legitimate and nonlegitimate sources of prescription medications can be tricky. This infographic illustrates the legitimate supply chain for prescription medications.
The Foundation partnered with the Food and Drug Administration (FDA) to host a public meeting on "Mitigating Risks from Human Xylazine Exposure" that explored real-world experiences and scientific evidence on emerging data trends. The meeting also examined concrete strategies for drug development and clinical research that directly support the mitigation and reduction of risks associated with human exposure to xylazine. Presenters and discussants included clinical and scientific experts, community and harm reduction organizations, academic researchers, and federal partners. The Foundation produced a report from the meeting that outlines actions needed to address xylazine exposure and the larger overdose crisis.
In June 2023, FDA issued its first psychedelics draft guidance for industry, Psychedelic Drugs: Considerations for Clinical Investigations, to provide general considerations to sponsors developing psychedelic drugs for treatment of medical conditions. While the guidance highlights some considerations for designing clinical trials with psychedelics to optimize the interpretability of results, many questions remain about the most appropriate way to address these challenges. The FDA Foundation convened “Advancing Psychedelic Clinical Study Design,” a public meeting to discuss the experience of scientists working with psychedelics in FDA-authorized clinical studies and drug development, considerations for psychedelics in clinical trial designs, and perspectives and current research in psychedelic clinical trials. The meeting also provided an overview of Psychedelic Drugs: Considerations for Clinical Investigations.
In recent months, families and individuals have reported increased challenges in accessing prescribed stimulant medications such as those used to treat Attention-Deficit/Hyperactivity Disorder (ADHD). The Foundation, in partnership with the FDA, has embarked on a new project to illustrate the factors that affect prescription stimulant availability.
The pathway to access these medications, classified as controlled substances, may seem straightforward with a relatively simple supply chain. In reality, the path of a prescription stimulant—from approval through manufacturing, distribution, and dispensing to the patient—has a number of challenging steps and requirements, any of which may slow down or interrupt the movement and accessibility of the medication. This preliminary image presents an illustrative but not exhaustive view of that complexity.
Buprenorphine is a safe and effective prescription medication used for the treatment of Opioid Use Disorder (OUD). Initiation of buprenorphine takes careful planning: patients must already be experiencing mild to moderate withdrawal symptoms or risk precipitated withdrawal. An increase in fentanyl in the illicit drug supply further complicates buprenorphine initiation and maintenance adding to the access challenge.
The Foundation, in collaboration with FDA and the Substance Abuse and Mental Health Services Administration, held a two-part virtual public meeting on “Considerations for Buprenorphine Initiation and Maintenance Care” to support efforts to develop products and approaches to treat OUD.
This report summarizes the discussions and presentations which explored solutions, innovative public health interventions and needed investments in research and product development to spur progress.
Drug overdose persists as a major public health issue in the U.S. Synthetic opioids, such as fentanyl and its analogs, are the primary driver of the increase in overdose deaths.
In March 2023, the FDA Foundation, in partnership with the Food and Drug Administration (FDA) and the Substance Abuse and Mental Health Services Administration (SAMHSA), hosted a virtual public workshop titled “Understanding Fatal Overdoses to Inform Product Development and Public Health Interventions to Manage Overdose” to examine the evolving context surrounding fatal overdoses and strategies to better manage overdose moving forward.
This summary report explores solutions and addresses regulatory considerations for future action and innovation.
The FDA Foundation, in collaboration with FDA and the National Institute on Drug Abuse (NIDA), hosted a virtual public workshop to discuss a practical research agenda toward treatment development for stimulant use disorder.
Meeting participants responded to a proposed practical research agenda that focuses on innovation in clinical trial design and candidate endpoints for the evaluation of potential treatments for stimulant use disorder.

Individuals inherently deserve services that promote health, regardless of whether they use drugs. Harm reduction is a set of proactive and evidence-based approaches that reduce the negative personal and public health impact associated with the use of alcohol and other drugs.
The FDA Foundation, working with FDA and several operating divisions within HHS, held two roundtables exploring community and clinical perspectives on fentanyl drug checking and screening. Community participants discussed their experiences using fentanyl test strips (FTS) and other drug checking methods, and clinician and researcher participants shared their perspectives on both clinical fentanyl testing and distribution of FTS to patients.
The FDA Foundation partnered with FDA’s Center for Drug Evaluation and Research to gain a deeper understanding of individuals’ experiences with substance use disorders (SUDs) – and specifically with SUD treatment. Our approach was to track individual treatment journeys to identify the specific challenges they faced and the supports they found helpful. Our goal is to use this ethnographic research to identify potential system improvements and engagement opportunities that can help inform FDA’s work in the prevention and treatment of SUDs. For this project, we looked at two specific issues: experiences exploring or using medication for opioid use disorder (MOUD), and trajectories of treatment for individuals who have pursued SUD treatment more than once.
Individuals exploring the use of medication to treat SUDs do not travel their paths alone. They are connected to networks of providers, peers, and family members who may have their own experiences, opinions, training, and expertise. Mapping individuals’ experiences led us to also map the experiences – and potential influences – of prescribing clinicians, non-prescribing clinicians such as counselors and therapists, and pharmacists. Individuals and these provider groups demonstrated generally positive views of MOUD and SUD treatment in general.
The Foundation teamed with FDA to host this virtual public meeting addressing some of the most frequently asked questions about access to naloxone, an antidote to opioid overdose.
Harm reduction specialists, physicians, pharmacists, and regulators explored the current landscape of naloxone availability, examined perceived barriers to naloxone access, and addressed questions posed by various groups on this subject. We then opened mics for nearly an hour of public comment to hear first hand from communities.
The Naloxone Economic View Summary Report explores the current distribution of naloxone in the U.S. and the potential economic impacts of a change in the prescription-only status of naloxone. The Foundation, in collaboration with FDA, analyzed data to estimate the amount of naloxone distributed in 2021. The report also includes findings from our expert roundtable with the payor community focused on understanding how a shift is the prescription status of naloxone might impact costs and consumer access.
The FDA Foundation convened, in conjunction with FDA’s Center for Drug Evaluation and Research, a roundtable on “Payor Perspectives on Substance Use Disorder Treatment.” The purpose of the roundtable was to hear key stakeholders’ perspectives on health insurance coverage of pharmacological treatments for SUDs, including how FDA approvals and labeling affect payors and insurance coverage.
Patient advocates, clinicians, payors, health economists, and federal leaders all participated. Attendees discussed the data framework insurance companies utilize in making coverage decisions for patients receiving substance use disorder treatments. The convening additionally generated real-world insight into how regulatory decisions are applied in ways that impact people’s daily lives.
The Reagan-Udall Foundation for the FDA, in collaboration with the U.S. Food and Drug Administration, hosted a hybrid public meeting titled Understanding Current Use of Ketamine for Emerging Areas of Therapeutic Interest. The meeting brought together clinicians, researchers, patient advocates, federal partners, and professional organizations to examine the evolving use of ketamine in clinical settings.
Discussions focused on emerging therapeutic applications, safety considerations, and the growing online promotion and access to ketamine. Presenters also reviewed the science behind ketamine’s pharmacology—including its approved use as an anesthetic and its potential antidepressant effects—and considered regulatory pathways, such as the supplemental new drug application (sNDA), for expanding approved indications. Insights from esketamine (Spravato®) development were explored as a potential model for future regulatory review.